Medical Device Quality Management System (QMS)
An audit-ready eQMS for medical device and MedTech companies.
Stay compliant and prepared for audits across ISO 13485, EU MDR, IVDR and FDA QSR.
A validated, cloud-based eQMS that simplifies compliance and reduces time to market.
ISO 13485:2016 Certified
Audit-ready eQMS for medical device companies
Stay compliant, in control and prepared for audits – across ISO 13485, EU MDR, IVDR and FDA QSR.
MedQdoc is a validated, cloud-based eQMS designed specifically for medical device and MedTech companies that need a structured, scalable quality management system – without unnecessary complexity or consultant dependency.
Stay compliant with EU MDR and IVDR regulations and maintain continuous audit readiness.
Why MedQdoc
MedQdoc is designed to support medical device and MedTech companies throughout the entire quality journey – from early development to audits, certification and global market access.
-
Always audit-ready
Maintain continuous compliance with ISO 13485, EU MDR, IVDR and FDA QSR through structured processes, traceability and real-time oversight. -
Simplified quality management
Replace fragmented documents and manual tracking with one integrated eQMS for documentation, risk management, CAPA and quality cases. -
Use what you already have
Migrate existing documentation or start from scratch – MedQdoc adapts to your organization, not the other way around. -
Built-in regulatory guidance
160+ ready-to-use templates mapped to regulatory requirements help you work efficiently and consistently. -
Designed for growth
Scale from early development to global market access while maintaining control and audit confidence.
Making your medical device eQMS journey simple to achieve
A user interface created with medical device compliance expertise – utilising pre-defined templates logically.
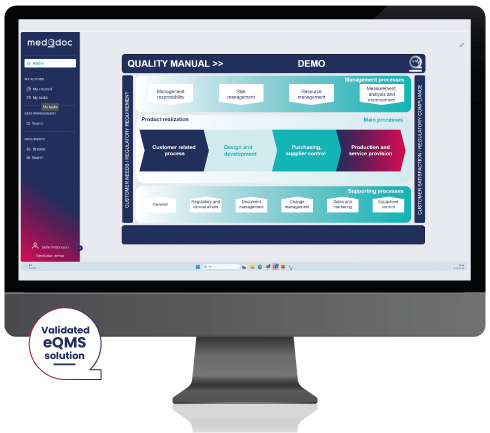
The MedQdoc Medical Device Quality Management System effectively applies procedures for:
- Quality Manual
- Management Responsibility
- Risk Management
- Resource Management
- Measurement, Analysis & Improvement
- Product Realization
- Customer related processes
- Design & Development
- Purchasing, Supplier Control
- Production & Service Provision
- Regulatory Affairs
- Document Management
- Change Management
- Equipment Control
Don’t let quality and regulatory requirements become a barrier to your medical device innovation
Enable your team by utilising MedQdoc eQMS, a medical device quality management system designed by medical device specialists. Request a consultative demonstration today >

Medical device focused

Designed by compliance experts

Validated SaaS platform

Over 160 templates

Simple step-by-step workflow

Ready-to-use / get started!

Cloud based flexibility

Simply tailorable if required
“Using MedQdoc’s framework gives me confidence that we will always
be audit-ready.”
Espen Westgaard, CTO, Aidee Health



