Efficient supplier management and control with MedQdoc's predefined workflows
Medical Device Supplier Management Functionality
The supplier management functionality in MedQdoc includes structured, ready-to-use workflows that streamline qualification, approval, and re-evaluation of suppliers. All activities are securely documented with electronic signatures and linked records such as certificates, audits, and CAPAs. Automatic audit trails and version control ensure that every supplier action remains traceable and compliant.
Workflow for Supplier Management
The MedQdoc Supplier Management workflow ensures structured, compliant, and traceable control of all supplier-related activities. Built within the validated MedQdoc eQMS, it supports ISO 13485, MDR, IVDR, and FDA QSR (21 CFR Part 820.50) requirements.
With role-based approvals, built-in risk classification, and automated reminders, MedQdoc helps you maintain approved supplier lists, manage certificates, and document every decision for audit readiness — all within a validated eQMS environment.

Key Benefits
- Centralized supplier qualification, evaluation, and performance tracking.
- Automated reminders for supplier re-evaluation and certificate renewals.
- Built-in risk classification and role-based approvals for controlled supplier management.
- Compliant with ISO 13485, MDR, IVDR, and FDA 21 CFR 820.50 requirements.
Regulatory Framework
MedQdoc Supplier Management supports international requirements for supplier control and purchasing processes:
- ISO 13485:2016 — Clause 7.4 (Purchasing)
Requires documented procedures for supplier evaluation, selection, and re-evaluation to ensure conformity of purchased product and services. - FDA 21 CFR 820.50 (Purchasing Controls)
Requires manufacturers to evaluate, select, and monitor suppliers and maintain approved supplier lists. - MDR/IVDR — Annex IX, Chapter I
Requires manufacturers to ensure supplier oversight for critical suppliers impacting device quality and regulatory compliance.
When to Use Supplier Management
- Onboarding new suppliers or service providers requiring qualification.
- Periodic re-evaluations or certificate renewals.
- Supplier-related nonconformities, CAPAs, or audit findings.
- Changes impacting supplier performance or compliance scope.
Want to explore how Supplier Management works in MedQdoc?
See detailed explanations and examples of automated supplier workflows, risk-based classifications, and best-practice forms below.
Quick access to your approved supplier list
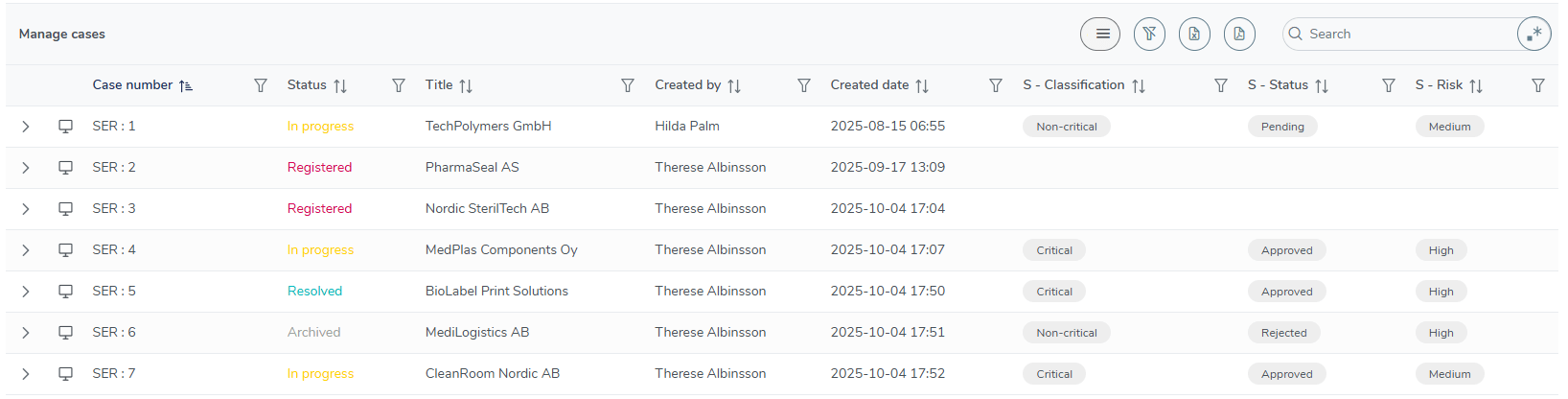
On the user-friendly MedQdoc dashboard, you’ll find your Approved Supplier List, giving you an instant overview of all suppliers — approved, pending, or rejected — along with their risk level and classification. Each supplier is managed as an individual case within MedQdoc, ensuring full traceability from registration to evaluation and re-approval. With just one click, you can filter, print, or export the list — making supplier oversight simple, transparent, and always audit-ready.
Pre-defined workflow for supplier registration
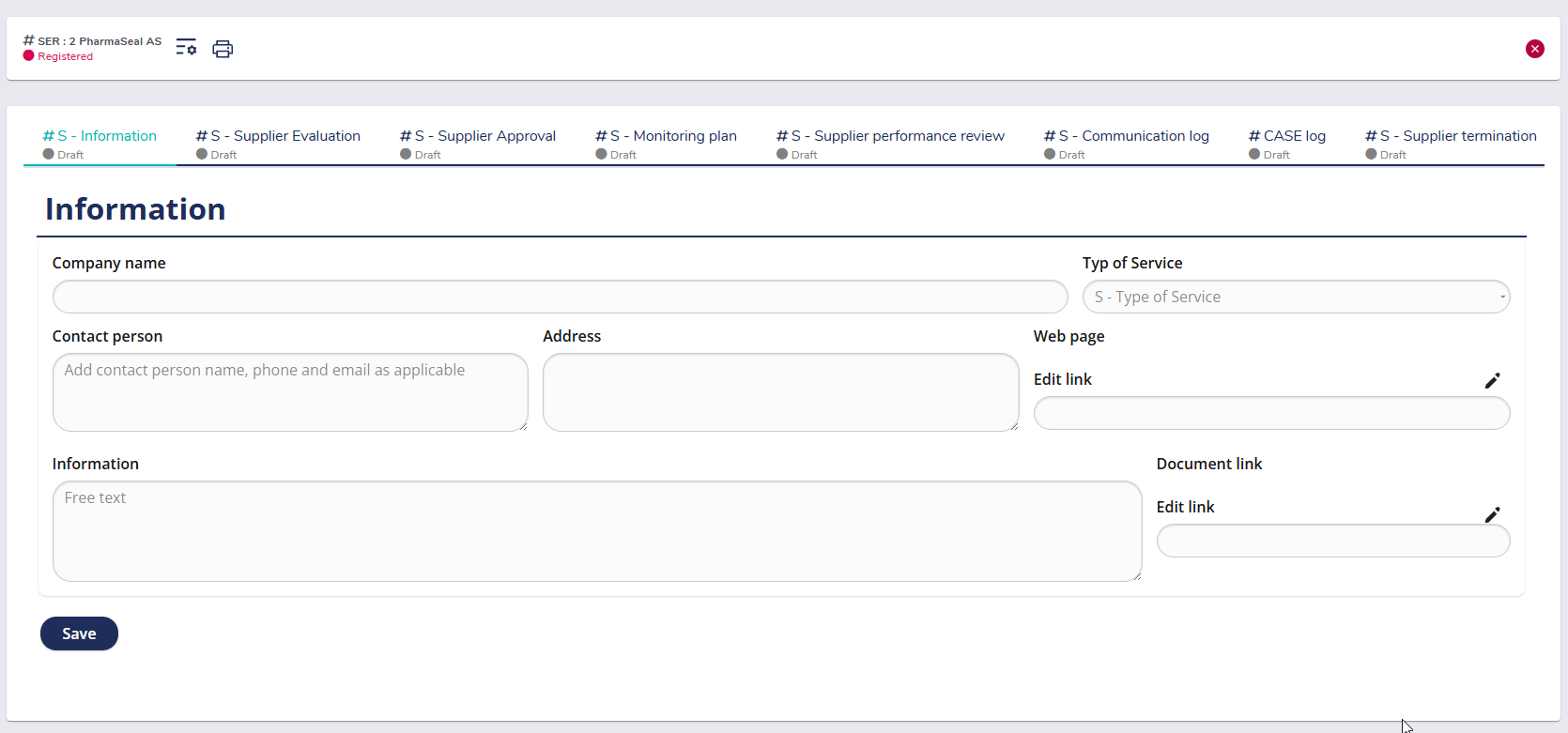
The predefined workflow in MedQdoc simplifies the way you register and classify suppliers. Each new supplier is created as an individual case, where company details, contact information, and service category are recorded in a standardized format. This ensures a consistent and compliant onboarding process aligned with ISO 13485 §7.4, providing a complete foundation for supplier evaluation and approval.
Supplier Evaluation – Structured and Risk-Based Assessment
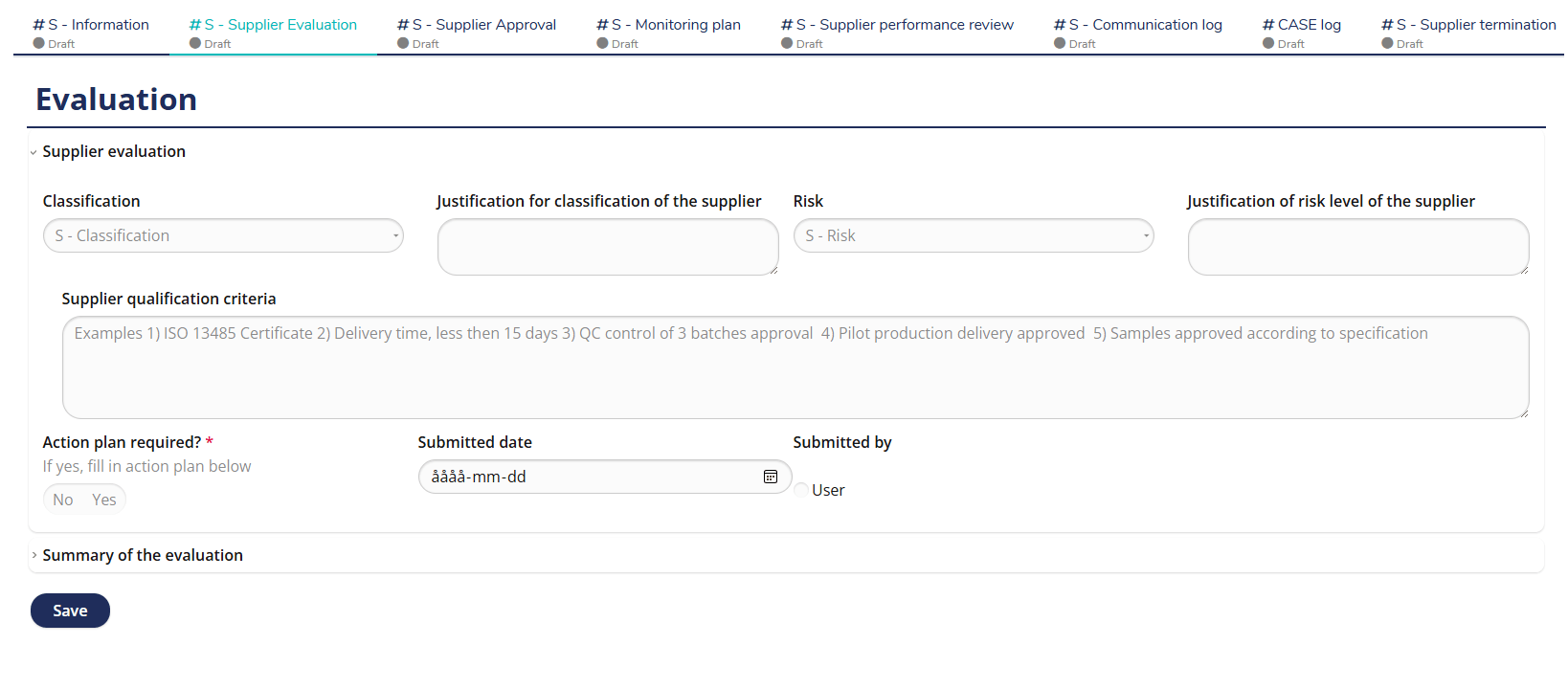
MedQdoc’s predefined Supplier Evaluation workflow helps you assess and qualify suppliers in a structured, risk-based manner. Using predefined evaluation criteria, you can document qualifications, risk levels, and justification for approval — all traceable and electronically signed. Automatic notifications remind users when evaluations are due, minimizing compliance risks and keeping your supplier records up to date. Fully aligned with ISO 13485 §7.4 and MDR supplier control requirements.
Well-structured supplier monitoring plan
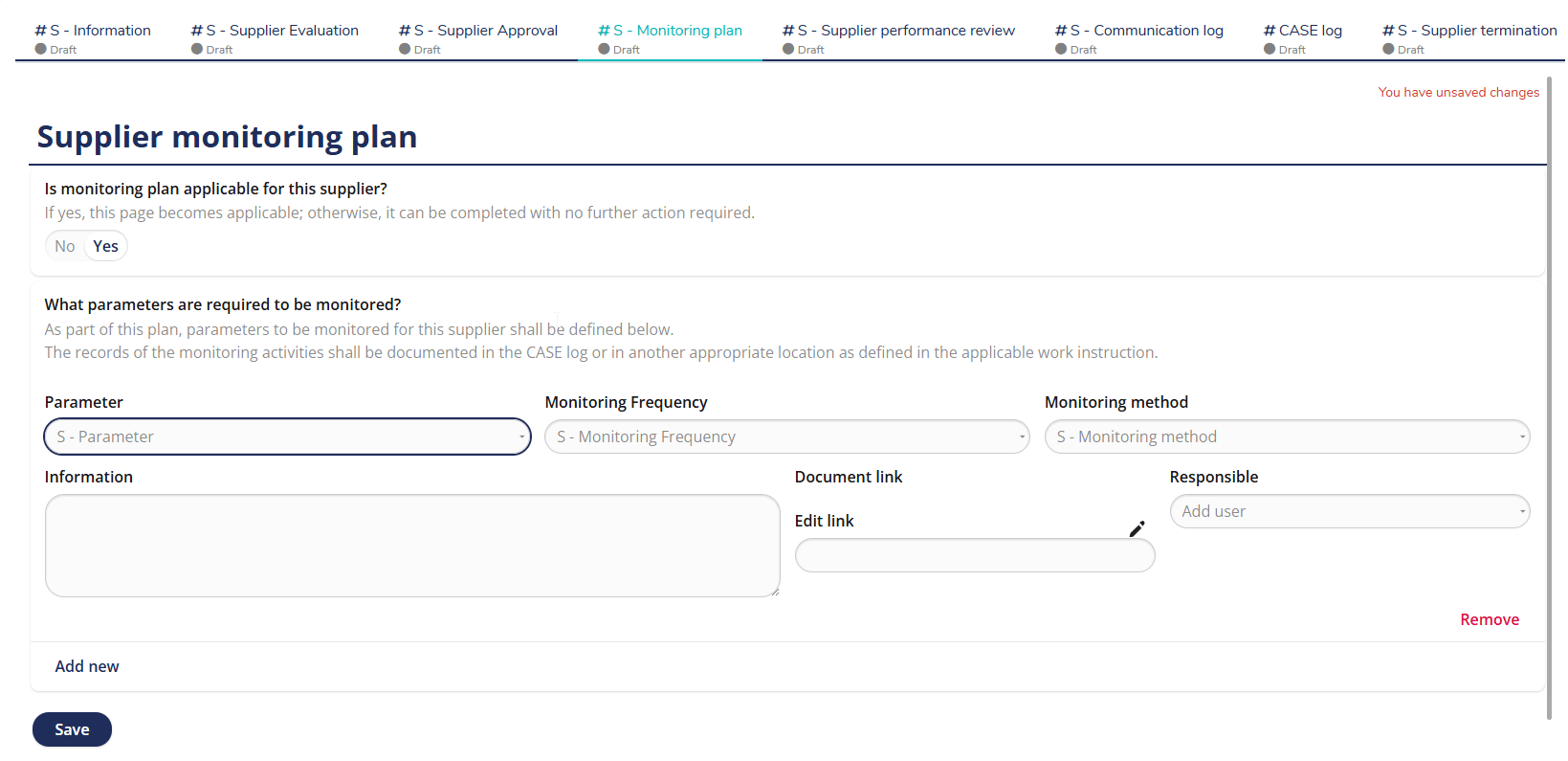
Maintain continuous oversight of supplier performance with MedQdoc’s Supplier Monitoring Plan. Define key parameters, responsible users, monitoring frequency, and methods — all in one central location. Automatic reminders help ensure supplier performance reviews are completed on time and documented according to your QMS procedures. Monitor supplier KPIs, actions, and follow-ups in a validated, audit-ready system.
Supplier Performance Review – Continuous evaluation with predefined criteria
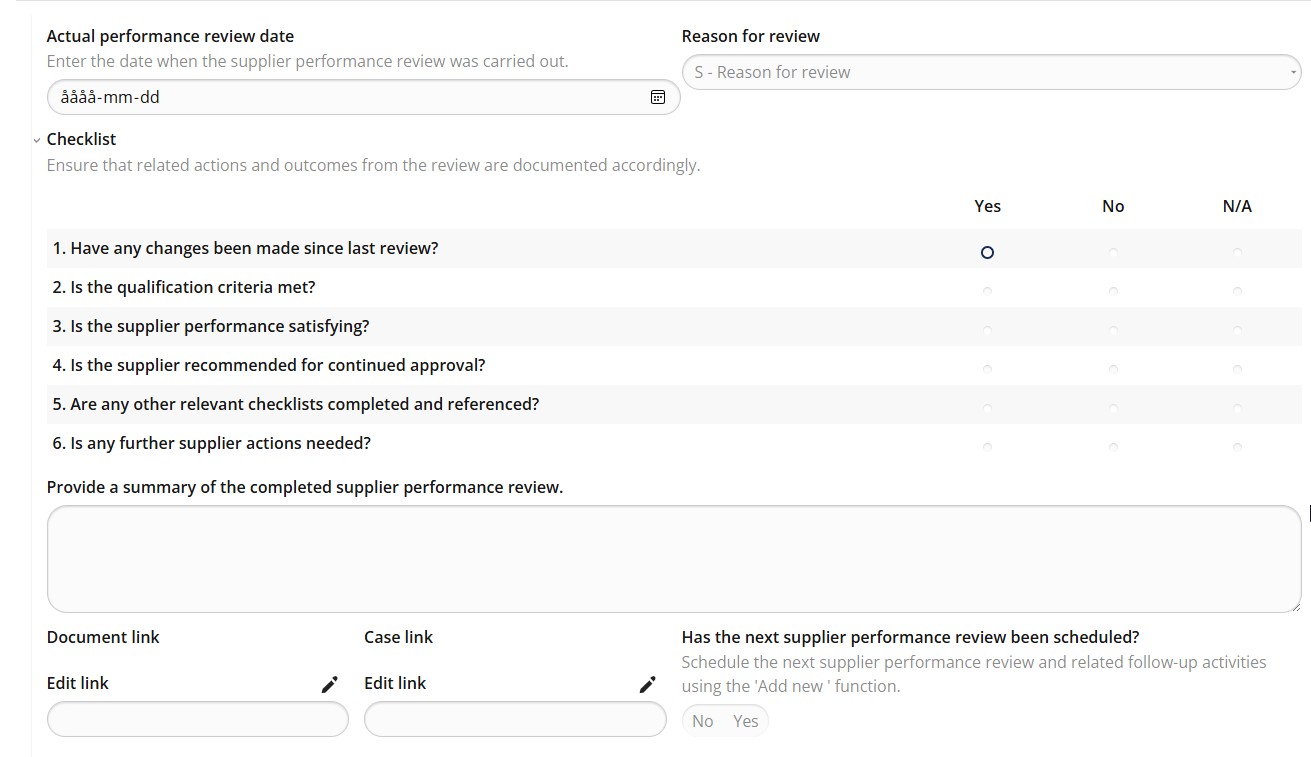
MedQdoc’s Supplier Performance Review workflow ensures that supplier evaluations are conducted consistently and on schedule. Predefined checklists guide users through every review, helping you verify supplier performance, delivery reliability, and quality results — all fully traceable within the system. Automatic notifications remind you when reviews are due, ensuring compliance with your QMS and regulatory requirements without missing critical deadlines. Certificates, agreements, and other supporting documents are securely stored and version-controlled — giving you a complete and validated supplier record from day one. MedQdoc’s document lifecycle then guides you through review and approval, ensuring full traceability and compliance throughout your supplier management process.
Supplier Questionnaire – Secure and Simple Supplier Information Collection

Simplify your supplier onboarding process with MedQdoc’s Supplier Questionnaire. Send a digital form to your suppliers and easily upload their completed information into MedQdoc, automatically linking it to the relevant supplier case. MedQdoc’s document lifecycle then guides you through review and approval, ensuring full traceability and compliance throughout your supplier management process.
MedQdoc medical device supplier management helps you to comply effectively with:
MDR / IVDR
ISO
13485
ISO
14971
QSR
(FDA)
CE-marking
process
21 CFR
Part 11
![]()
Designed by medical device quality and regulatory compliance experts.
![]()
Includes over 160 QMS and MDR/IVDR technical documentation templates for medical device compliance.
![]()
Intuitive and user friendly; simple and effective ISO 13485 document control.
![]()
MedQdoc is loved by auditors.
“I would absolutely recommend MedQdoc to other companies in the medical device world. Without a doubt, using MedQdoc has been a key factor in the success of our ISO 13485 certification process.”
Kristian Nisja, QA & HSE Manager,
NORBIT EMS Selbu

MedQdoc has extensive functionality in all of the QMS areas below, please talk to the MedQdoc team for specific queries that you may have:
Quality Management System
Change Management
Supplier Management
Training Management
CAPA
Non- Conformities
Complaint Handling
Customized Case Flows
Template Management
Technical Documentation



