Medical device document control for QSR and ISO 13485
Intuitive, streamlined and automated medical device document control
Simple and streamlined document control in a medical device QMS built to provide effective collaboration and assured compliance with QSR and ISO 13485. Designed by medical device regulatory compliance and QMS experts, MedQdoc is designed to enable intuitive medical device document control for QSR and ISO 13485 to accelerate the journey to compliance.
MedQdoc is validated and developed by an ISO 13485:2016-certified company with built in functionality for companies to comply with MDR, IVDR, QSR, ISO 13485, ISO 14971, ISO 9001 and ISO 27001
The medical device document control functionality within MedQdoc includes:
Quick access to your medical device quality management systems
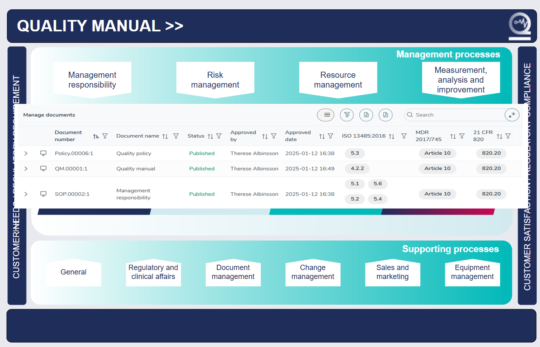
Get a complete, real-time overview of all your processes and related documentation right from the MedQdoc start page.
Your document My tasks in a structured logical way
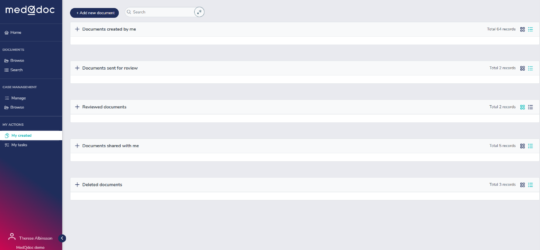
In the MedQdoc My Tasks page, you can view all actions assigned to you for each document — guiding you through every step of the review and approval process.
Quickly set correct references to the relevant standard or legislation
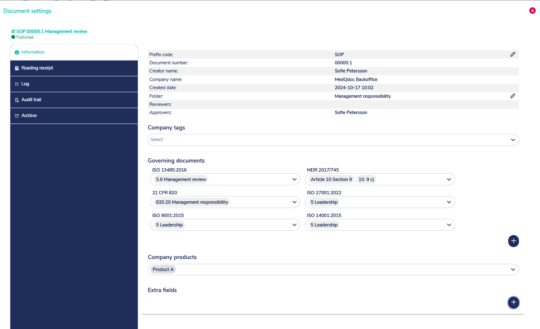
MedQdoc contains preconfigured lists with references to ISO 13485, ISO 9001, ISO 27001, QSR, 21 CFR Part 11 and it is easy to set references for MDR and IVDR.
Use the built-in templates to get a head start creating your QMS
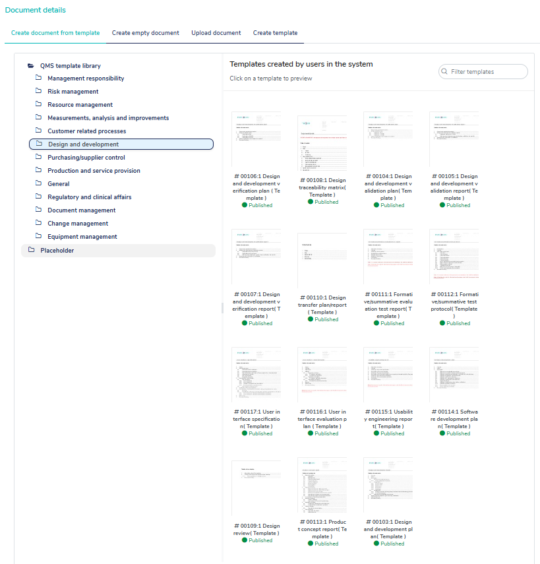
MedQdoc includes templates aligned with ISO 13485, ISO 14971, QSR, MDR 2017/745, and IVDR 2017/746 — helping you build your QMS and technical documentation efficiently and in compliance.
Create and edit with the built-in editor - No need for Office or check-in/check-out
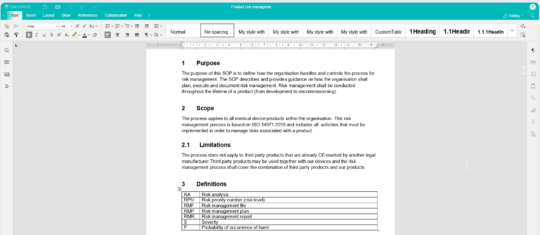
MedQdoc includes a built-in editor, removing the need for external Office tools. Edit documents directly from any computer, tablet, or phone using a browser. Keeping the document within the system at all times ensures that every action is logged and traceable.
Document meta data clearly visible in the header of a document
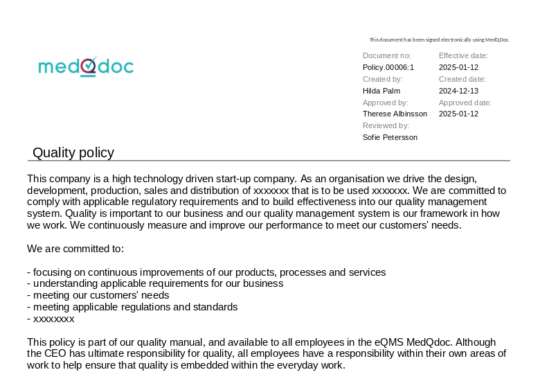
When accessing a document in MedQdoc, key metadata such as version, creator, reviewer, approver, and relevant dates are clearly visible — ensuring clarity and compliance with QSR and ISO 13485 document control requirements.
Flexible access control to your QMS
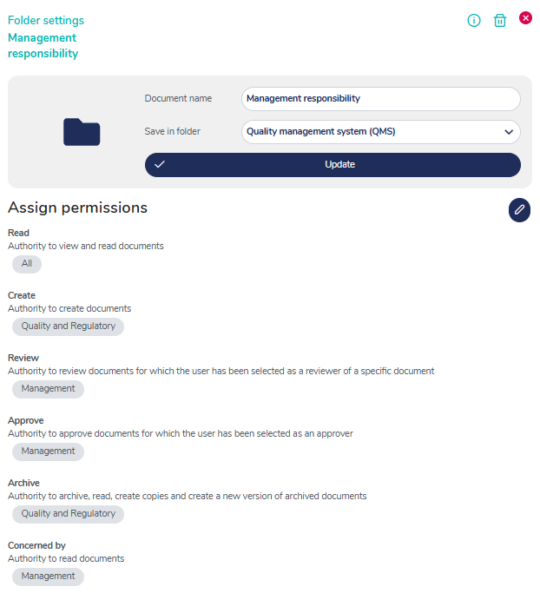
Control access and authorization across your QMS at the individual, group, department, or company level — giving each user exactly the access they need.
Reviewal and approval process with 21 CFR Part 11 compliance
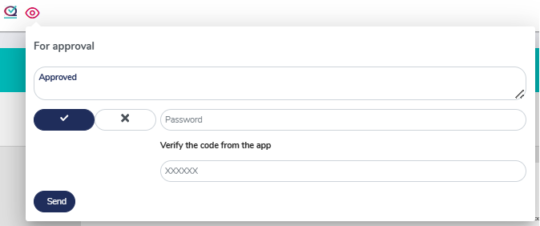
MedQdoc features a clear and structured review and approval process with secure password authentication and optional two-factor verification for reviewing or approving documents.
Easily compare changes between different versions of a document
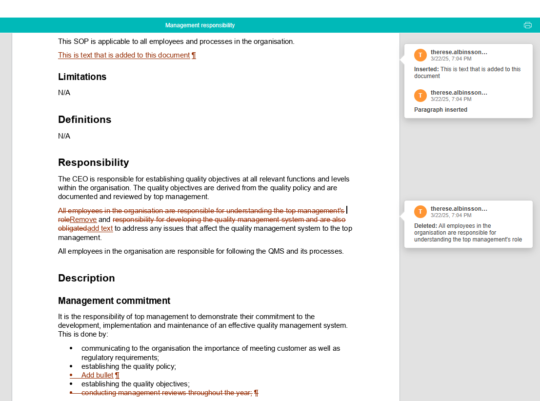
Compare any document version in MedQdoc to see exactly what changed, when, and by whom.
Complete audit trail with timestamps for clarity
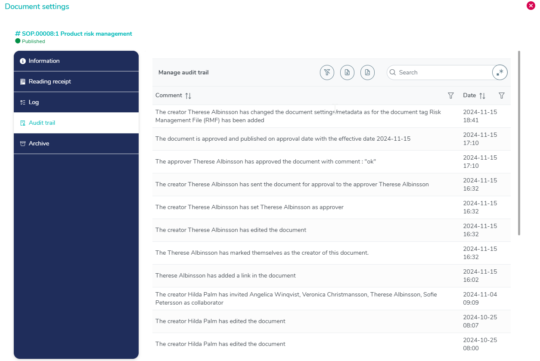
The audit trail captures all document changes with details on what was modified, by whom, and when. Each entry is securely timestamped and meets 21 CFR Part 11 requirements for electronic records and signatures.
Flexible document connections using tags

Use the included tags, or create your own, to connect related QMS documents across different structures — for example, all files in your Risk Management File, Usability Engineering File, DMR, or DHR.
Create updateable document lists
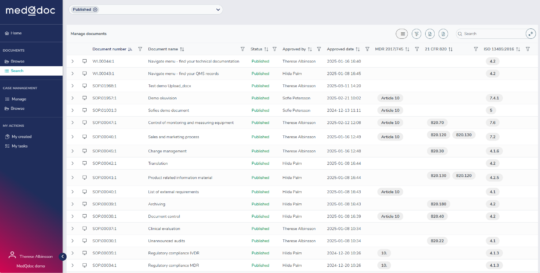
Easily create dynamic document lists using any metadata and save them for future updates. For example, generate a complete QMS document list with references to ISO 13485 and QSR — always current and audit-ready A dream for an auditor!
MedQdoc medical device document control helps you to comply effectively with:
MDR / IVDR
ISO 13485
ISO 14971
QSR (FDA)
CE-marking process
21 CFR Part 11
![]()
Designed by medical device quality and regulatory compliance experts.
![]()
Includes over 160 QMS and MDR/IVDR technical documentation templates for medical device compliance.
![]()
Intuitive and user friendly; simple and effective ISO 13485 document control.
![]()
MedQdoc is loved by auditors.
“One of the top reasons we chose the system was because it was created by regulatory experts, which seems to be unique in the market.” Malin Nilsson, CEO, Inossia

MedQdoc has extensive functionality in all of the QMS areas below, please talk to the MedQdoc team for specific queries that you may have:



