Empower employee growth with MedQdoc’s streamlined training management solution
Easily manage and monitor your employees’ training requirements and progress with MedQdoc’s medical device training management system. Our comprehensive solution offers training templates, dedicated training spaces, and a straightforward training tracker, ensuring your workforce stays up to date with the necessary skills and qualifications they need. Simplify training management, enhance compliance, and empower team development with our user-friendly eQMS platform for MedTech companies.
MedQdoc is validated and developed by MedQtech, an ISO 13485:2016-certified company. The system supports compliance with MDR, IVDR, QSR, ISO 13485, and ISO 14971—helping medical device manufacturers maintain full regulatory control and training traceability.
Workflow for Training Management
The MedQdoc Training Management workflow ensures structured, compliant, and traceable control of all employee training activities. Built within the validated MedQdoc eQMS, it supports ISO 13485, MDR, IVDR, and FDA QSR (21 CFR Part 820) requirements.

Key Benefits
- Centralized training records and competence tracking.
- Automated reminders and retraining notifications.
- Linked to CAPA, Change, and Document Management modules.
- Compliant with ISO 13485 and FDA 21 CFR 820.25.
Regulatory Framework
MedQdoc Training Management supports ISO and FDA expectations for competence and training:
- ISO 13485:2016 — Clause 6.2 (Human resources)
Requires personnel competence, training needs determination, training provision, and effectiveness evaluation. - FDA 21 CFR 820.25 (Personnel)
Requires appropriate education, background, training, and experience; training records must be maintained. - MDR/IVDR — Article 10
Requires manufacturers to ensure adequate competence and training proportionate to assigned tasks.
When to Use Training Management
- New hires or role changes requiring documented competence.
- Updated procedures or product changes triggering retraining.
- Audit findings or CAPA actions requiring targeted training.
Want to explore how Training and Education records works in MedQdoc?
See detailed explaination and examples of automated workflows and best-practice forms below.
Structured training process

MedQdoc provides a structured process for documenting and managing employee competence and training — from identifying training needs to recording completed sessions and maintaining full ISO 13485 compliance.
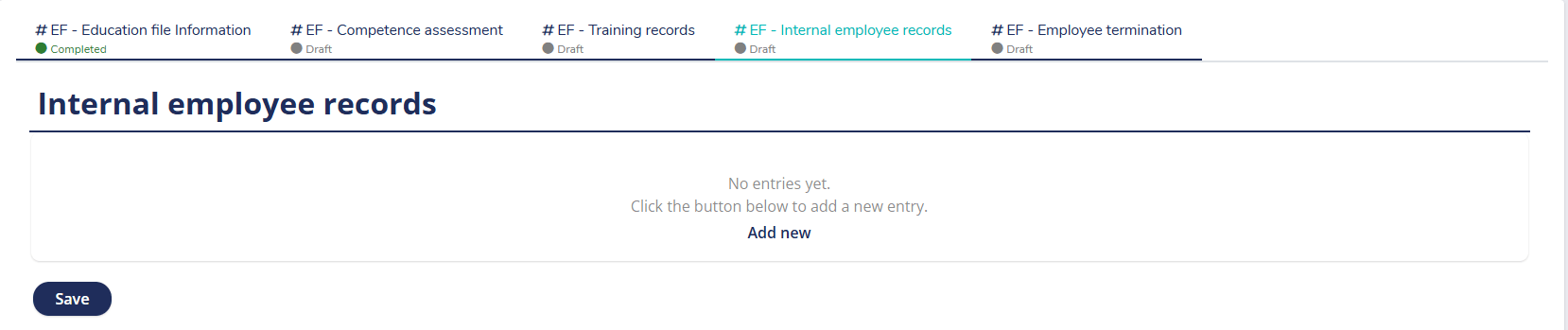
Ready-to-use form for registering training sessions

MedQdoc helps you easily register and track employee training records — from ISO 13485 courses to MDR 2017/745 examinations and Lead Auditor certifications — all fully traceable and compliant.
Individual training record for self-evaluation
Automatically notify the trainees and let them evaluate the training effectiveness. The trainer is automatically notified for instant follow-up.
Streamlined education folder structure

Organise and locate your educational materials easily with MedQdoc’s pre-defined folder structure, saving time and enhancing productivity.
Controlled access to training files
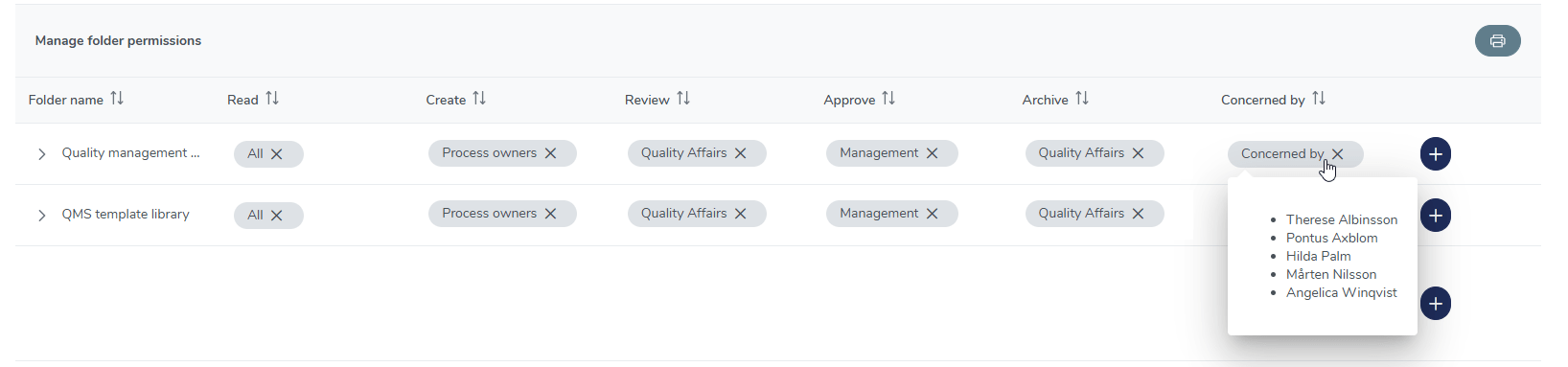
Safeguard your training materials and ensure that files are accessed only by those who need them to maintain compliance and confidentiality.
Training tracking and notification
![]()
Relevant personnel are promptly notified when new training documents or standard operating procedures become available. Receive read-receipts and track training completion to stay on top of training requirements.
Enhanced search functionality
MedQdoc’s powerful search functionality allows for quick and accurate retrieval of relevant training materials, supporting efficient onboarding and ongoing learning initiatives.
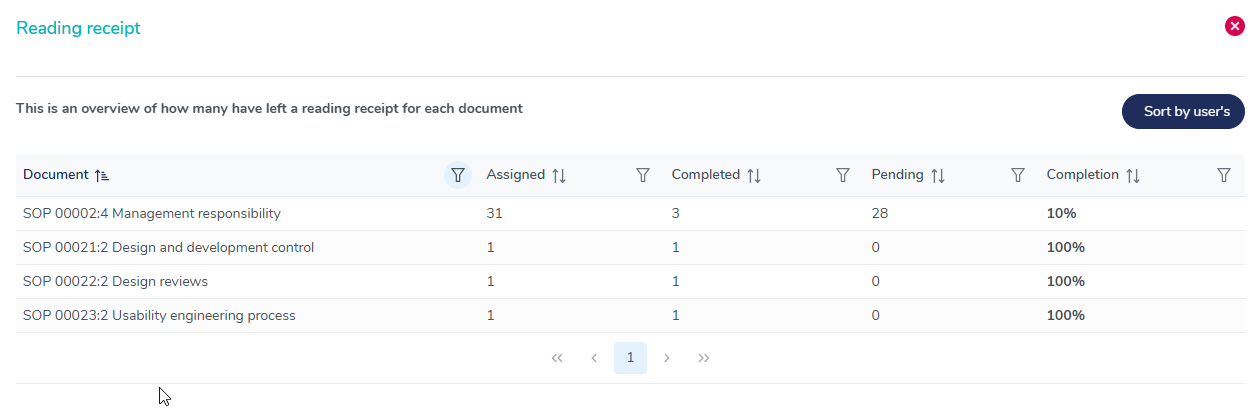
User training completion overview
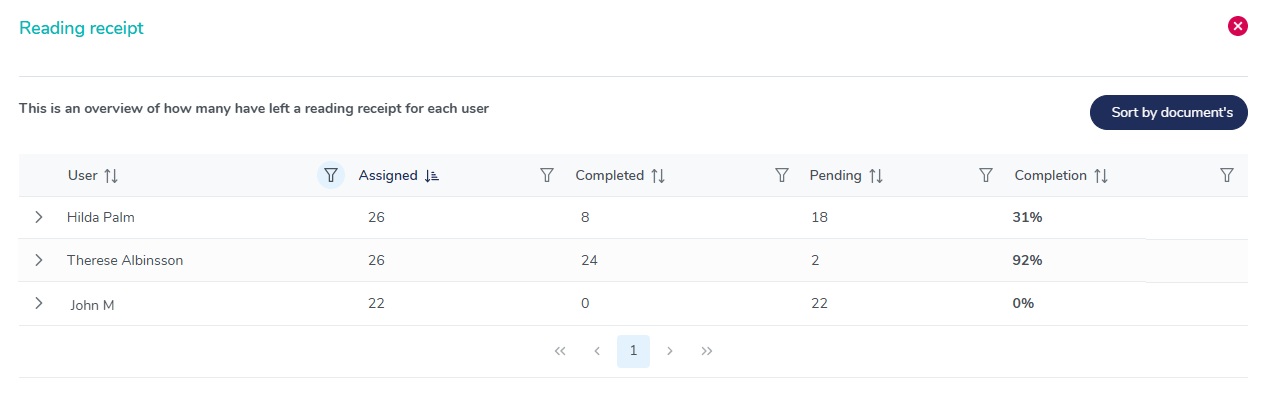
Gain full visibility into employee training completion rates with MedQdoc’s reading receipt overview. Instantly track pending acknowledgements, monitor training effectiveness, and ensure full traceability for audits and compliance.
Audit trail of case and document
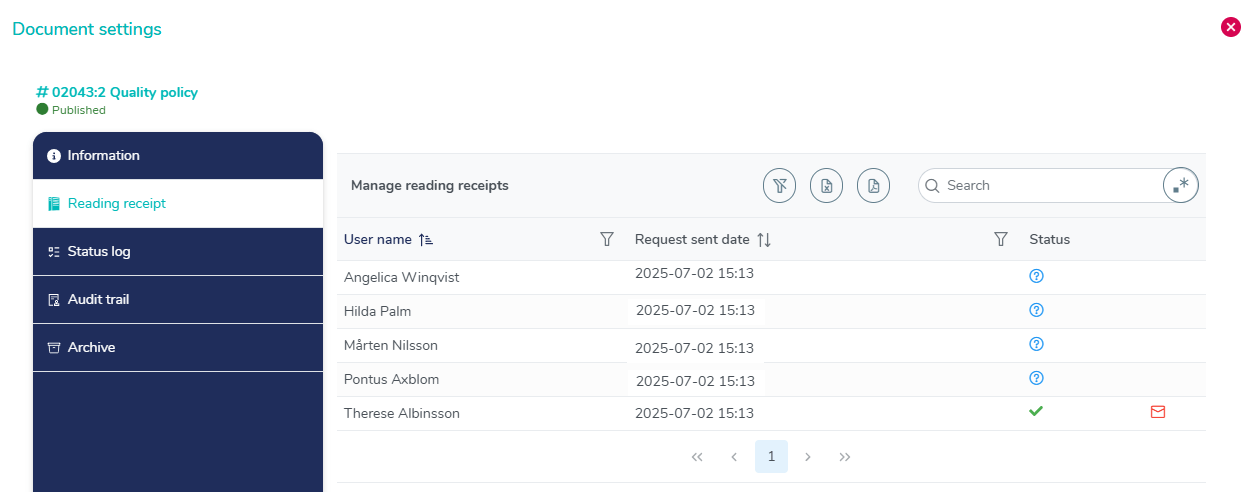
Benefit from transparent audit trails and advanced tagging functions to manage and keep track of all training documentation, ensuring accountability and compliance.
User training sign-off for compliance
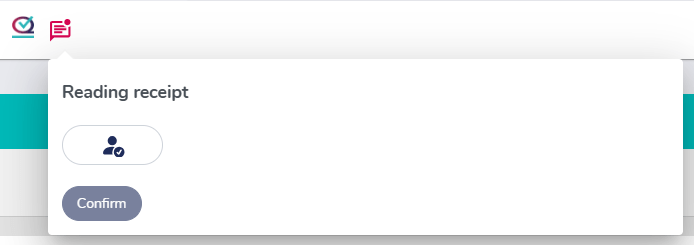
Audit readiness! Capture electronic signatures and acknowledgements from trainees to ensure that they have reviewed and understood the training content, forming a robust training validation system.
MedQdoc medical device training management helps you to comply effectively with:
MDR / IVDR
ISO
13485
ISO
14971
QSR
(FDA)
CE-marking
process
21 CFR
Part 11
![]()
Designed by medical device quality and regulatory compliance experts.
![]()
Includes over 160 QMS and MDR/IVDR technical documentation templates for medical device compliance.
![]()
Intuitive and user friendly; simple and effective ISO 13485 document control.
![]()
MedQdoc is loved by auditors.
“One of the top reasons we chose the system was because it was created by regulatory experts, which seems to be unique in the market.”
Malin Nilsson, CEO, Inossia

MedQdoc has extensive functionality in all of the QMS areas below, please talk to the MedQdoc team for specific queries that you may have:
Quality Management System
Change Management
Supplier Management
Training Management
CAPA
Non- Conformities
Complaint Handling
Customized Case Flows
Template Management
Technical Documentation



