Manage changes easily with MedQdoc’s automated change management workflows
Effectively capture and manage changes with MedQdoc’s structured, traceable and automated workflows. The platform simplifies change management to ensure a seamless workflow for tracking modifications, approvals and implementation. Gain complete visibility over the change management process with our user-friendly system, empowering you to promote efficiency, compliance and error prevention throughout your organisation.

Workflow for Change Management
The MedQdoc Change Management workflow provides a structured, compliant, and fully traceable process to control and document changes throughout their lifecycle. Whether related to products, processes, suppliers, or documentation, MedQdoc ensures that every change follows a validated, auditable path aligned with ISO 13485, EU MDR (2017/745), and FDA QSR (21 CFR Part 820).
The following stages are part of the MedQdoc Change Management lifecycle, ensuring traceability, accountability, and visibility at every step:
- Initiation — A change request is logged, categorized (e.g., document, product, process), and evaluated for scope and impact.
- Impact Assessment — Risks, affected documents, and dependencies are analyzed to determine regulatory, quality, or operational implications.
- Planning — Change actions, responsible owners, due dates, and related CAPA or risk controls are defined and approved.
- Implementation — Approved changes are executed; updated records, SOPs, and training activities are attached for traceability.
- Verification — Changes are reviewed and validated to confirm compliance, effectiveness, and documentation accuracy.
- Closure — The change record is formally approved and archived, ensuring a complete audit trail and QMS compliance.
How it works
Each step can be saved as a draft or marked as complete to move the workflow forward. Once a step is completed, the Change Request automatically transitions to the next defined stage — maintaining structure, compliance, and control. Some steps may also include formal review or approval phases, depending on your organization’s configuration and role-based permissions.
Book a MedQdoc DemoRegulatory Framework
MedQdoc Change Management supports both ISO and FDA requirements for documented change control processes:
- ISO 13485:2016 — Clauses 4.1.4, 4.2.4, 4.2.5, and 7.3.9
Requires control of QMS documentation, product design changes, and process modifications through a defined and documented procedure. - FDA 21 CFR Part 820.30 and 820.40
Requires manufacturers to review, approve, and document changes affecting product design, processes, or documentation before implementation.
When to Use Change Management
- Product, process, or documentation updates that impact regulatory compliance or quality records.
- Changes triggered by CAPA, audit findings, or post-market surveillance outcomes.
- Supplier-related changes requiring evaluation and approval under MDR and ISO 13485.
- Internal process improvements, workflow optimization, or system upgrades within the QMS.

Key Benefits of Change Management in MedQdoc
- Centralized control of all QMS-related changes with complete traceability.
- Integrated links to CAPA, Risk Management, and Document Control modules.
- Compliant with ISO 13485:2016 and FDA 21 CFR 820 / QMSR requirements.
- Automated reminders, approvals, and version control for efficient change execution.
- Audit-ready documentation demonstrating strong governance and regulatory alignment.
Want to explore how Change Management works in MedQdoc?
See detailed examples of automated workflows and best-practice forms below.
Streamlined stepwise workflow
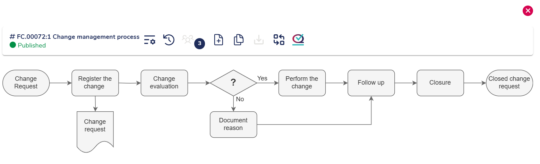
MedQdoc’s structured and stepwise workflow aligns with best practices and industry standards, helping you document changes more effectively and enabling compliance.
Built-in best practice change forms

Simplify change documentation with built-in forms that align with best practices. Record changes effortlessly, eliminating the need to start from scratch and ensuring adherence to industry standards.
Validated workflows
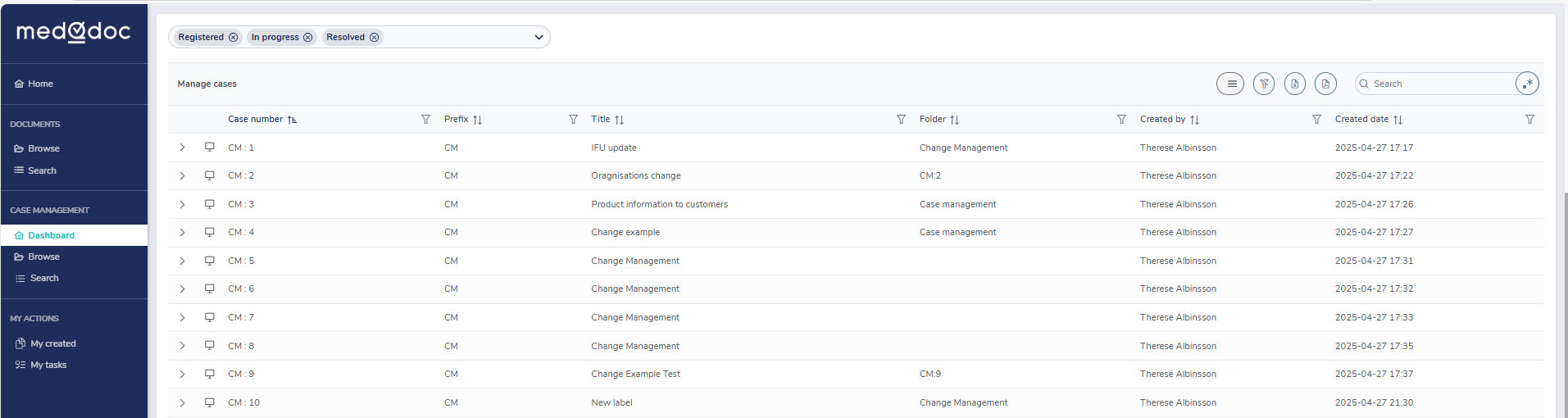
Have complete confidence and peace of mind knowing that your workflow follows validated protocols and supports compliance requirements.
Straight-forward data export
![]()
Export data in various formats to facilitate collaboration with external parties. Share information easily outside of the software, allowing stakeholders to review and provide feedback in their preferred format.
Trending and Data-driven analysis
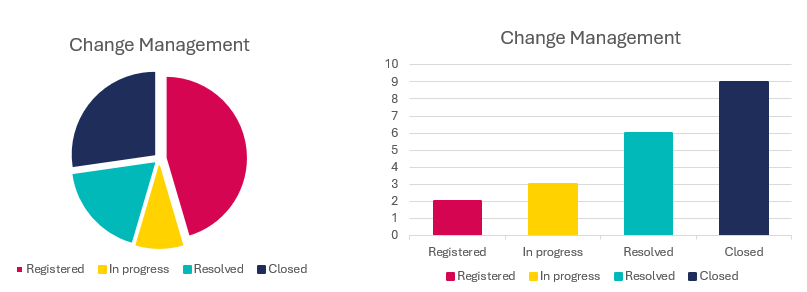
Gain valuable insights by analysing changes throughout the project. Track the progress of changes, and monitor the number of initiated and closed changes to make informed decisions based on real-time data.
Adaptable workflows for any organisation
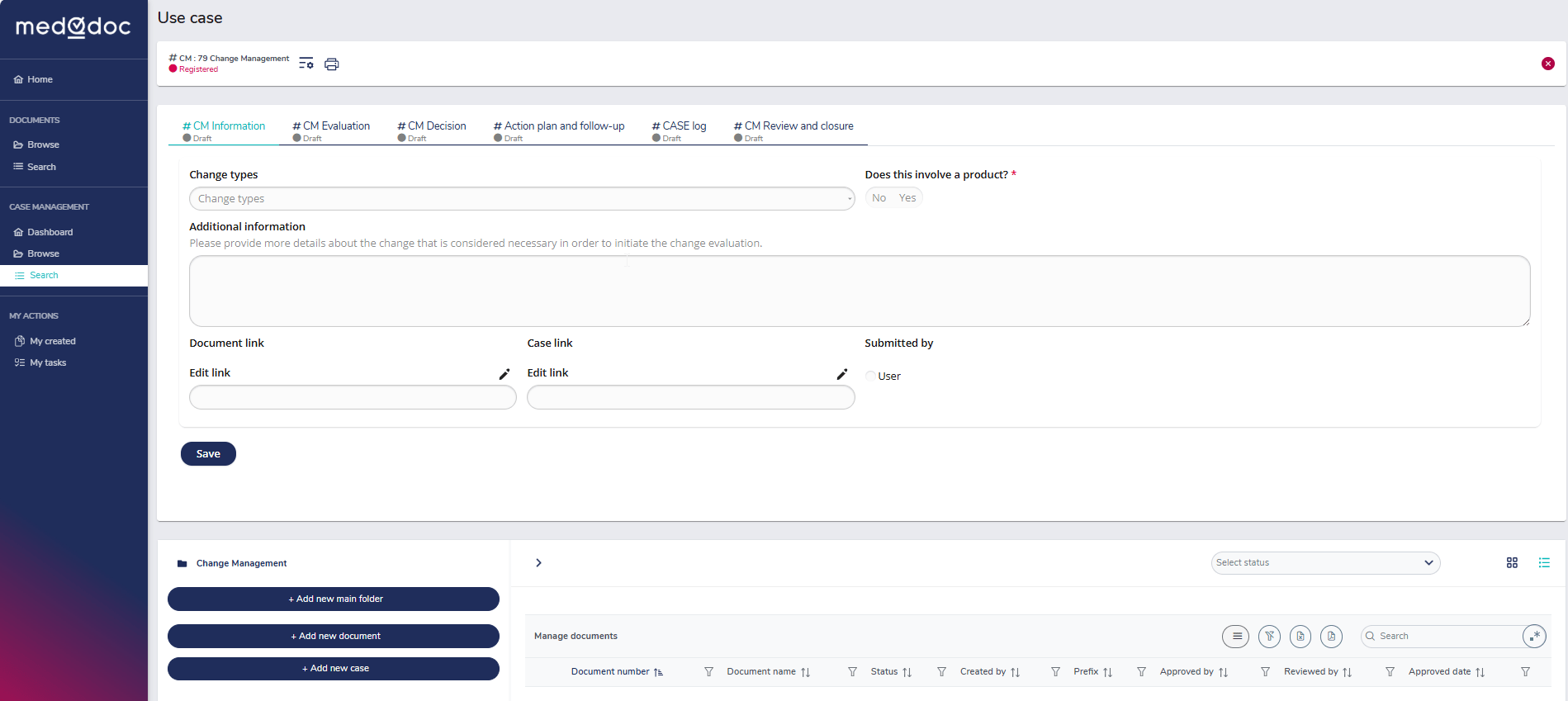
Customise and adapt MedQdoc’s workflows with complete flexibility to align with your company’s specific requirements and processes.
Document control for effective change management guidance
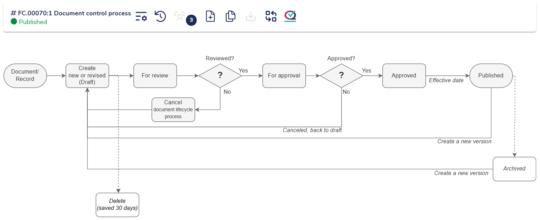
Maintain control over your documents to guide change management processes effectively. MedQdoc provides a consistent and structured way of working, eliminating the need to start from scratch with each change.
Powerful search functionality

Easily search and retrieve change-related information for daily operations and audits using MedQdoc’s intuitive search functionality. Locate specific change records based on criteria such as registration date, status or responsible persons.
MedQdoc medical device change management helps you to comply effectively with:
MDR / IVDR
ISO 13485
ISO 14971
QSR (FDA)
CE-marking process
21 CFR Part 11
![]()
Designed by medical device quality and regulatory compliance experts.
![]()
Includes over 160 QMS and MDR/IVDR technical documentation templates for medical device compliance.
![]()
Intuitive and user friendly; simple and effective ISO 13485 document control.
![]()
MedQdoc is loved by auditors.
“One of the top reasons we chose the system was because it was created by regulatory experts, which seems to be unique in the market.” Malin Nilsson, CEO, Inossia

MedQdoc has extensive functionality in all of the QMS areas below, please talk to the MedQdoc team for specific queries that you may have:



